Next-Generation Antibody Engineering for Alzheimer’s Disease
Alzora Bio is developing next-generation antibody engineering approaches designed to maintain efficacy while improving safety, expanding patient eligibility, and enabling real-world adoption.
About Us
Alzora Bio is a research group based at the University of Southampton developing next-generation antibody approaches for Alzheimer’s disease.
Our work focuses on enhancing the safety profile of amyloid-targeting therapies while maintaining clinical efficacy, with the aim of expanding the treatable patient population and improving real-world adoption.
As part of the ICURe Explore programme, we are engaging with industry stakeholders to understand clinical, commercial, and partnership opportunities for advancing safer Alzheimer’s therapies.
Help Shape the Future of Alzheimer’s Treatment
We are seeking insights from pharmaceutical and biotech professionals to better understand development priorities, safety considerations, and partnership opportunities in Alzheimer’s treatment.
Engineered Antibody Precision
Our approach uses targeted antibody engineering to selectively engage certain Fc receptors, enabling effective amyloid clearance with improved safety.
Optimised Immune Engagement
By refining how antibodies interact with Fc-receptors, we aim to minimise neuroinflammation and reduce the incidence of ARIA-related effects seen in current therapies.
antibody structure
Strategic Applications & Value Drivers

Expanding Market Opportunity
Safety limitations currently restrict anti-amyloid antibody therapies to a small subset of patients. Our approach is designed to improve tolerability, enabling access to a significantly broader patient population.
Opportunity: Up to 3x expansion in eligible population
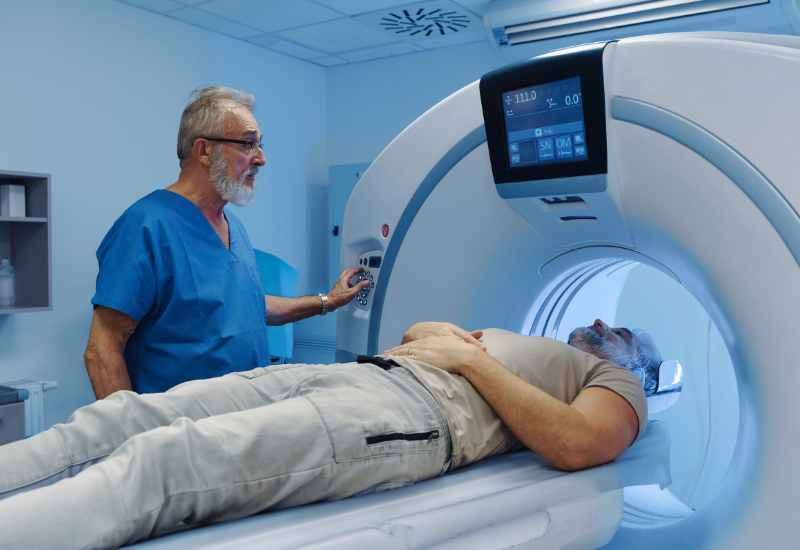
Enabling Scalable Delivery
Intensive MRI monitoring presents a key barrier to adoption and scalability. By reducing safety-related risks, our approach may lower monitoring requirements and support more efficient deployment across healthcare systems.
Opportunity: Reduced system burden & improved scalability

Unlocking Pipeline Potential
A safer antibody profile may enable earlier intervention, improved dosing strategies, and the re-evaluation of previously limited or discontinued therapeutic candidates across development pipelines and clinical programmes.
